Overview
Cell therapy research is a rapidly advancing field. Multiple cell therapies are now commercially available across the world to treat certain hematological disorders. A plethora of clinical trials for investigational therapies addressing a wide range of diseases like cancer, autoimmune disease and genetic disorders are using autologous or allogeneic cells with rapidly evolving cell engineering and editing approaches. A common theme in cell therapy research is the need to understand cell phenotypes and heterogeneity at every step of the process, including characterization and development of predictive biomarkers and cells isolated from patients as input for cell therapy manufacturing.
Flow cytometry is a powerful tool to characterize cell phenotypes to define critical quality attributes for cell therapy products such as cell viability, identity, potency and purity. Flow cytometry assays are performed at different stages during the development and manufacturing of cell therapy products. Assays are often transferred between multiple manufacturing sites for the final release and QC of therapeutic products.
Flow cytometry is a powerful tool to characterize cell phenotypes to define critical quality attributes for cell and gene therapy products such as cell viability, identity, potency and purity. Flow cytometry assays are performed at different stages during the development and manufacturing of cell therapy products. Assays are often transferred between multiple manufacturing sites for the final release and QC of therapeutic products.
BD Biosciences is your global partner in single-cell analysis. We provide extensive scientific resources and tools that enable high-dimensional analysis, automation and standardization because we are committed to partnering with you so that you can advance your cell therapy research programs by delivering high-quality solutions that support your 21 CFR Part 11 compliance.
Cell Therapy Development Journey
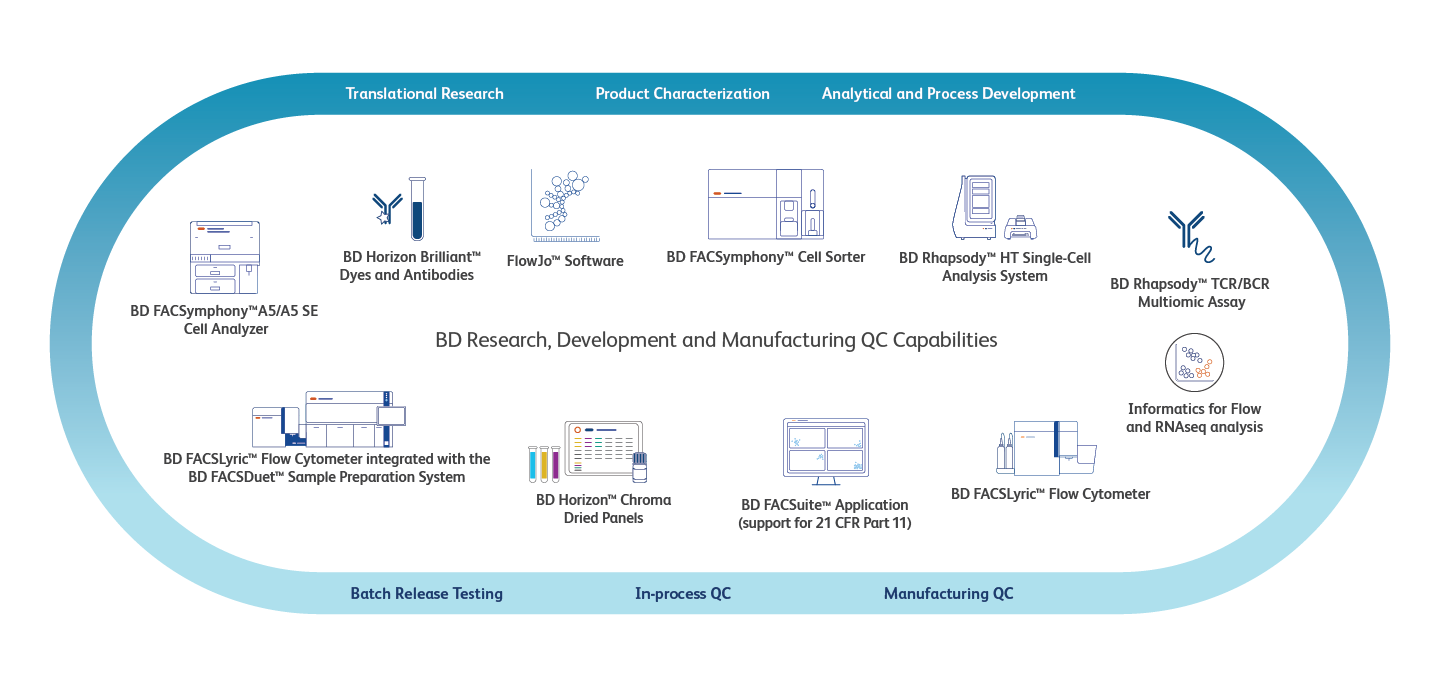
Research & Development
During the early stages of cell therapy research, a critical priority for process and product development is to characterize starting material and understand the biology, cell phenotype and function while maximizing data output. Our flexible innovative single-cell multiomics platform and high- dimensional flow cytometers allow in-depth characterization of cell function and research for the development of predictive biomarkers.
Manufacturing QC
In cell therapy manufacturing QC, automated and easy to use flow cytometers, reagents produced in ISO 13485:2016 certified facilities required to follow Medical Device Current Good Manufacturing Practice (cGMP)*, software features relevant to 21 CFR Part 11 and lab information systems are crucial for product release in accordance with GMP guidelines by the European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA). All user-defined flow cytometry assays used for manufacturing QC release of product must but validated and verified by the user. The user must comply to applicable local regulations and obtain appropriate Health Authority approval as necessary for their intended use.
Optimizing manufacturing processes to reduce complexity and improve scalability requires use of automated technologies. Flow cytometry solutions that are standardized across instruments allow data comparison and consistency of results across manufacturing sites and allow seamless tech transfers.
*as stated in the Quality System Regulations (QSR) [U.S.Food and Drug Administration Reference 21CFR 820].
The BD Rhapsody™ HT Single-Cell Analysis System enables single-cell capture with reliable and reproducible results with minimal site-to-site variability making it suitable for translational, multi-center, multi-cohort studies. One readout can be used to analyze both protein and mRNA, reducing experimental time and increasing information derived from a single experiment. BD® AbSeq Antibody-Oligonucleotide Conjugates allows high-parameter protein expression data simultaneously with single-cell RNA-Seq data to significantly improve your understanding of individual cells. For example, targeted panels like the BD Rhapsody™ TCR/BCR Multiomic Assay Kit allow protein and whole transcriptome mRNA analysis to comprehensively understand immune cell function. Using genomic and proteomic information from single-cell analysis, you can focus on cells of interest for research and a deeper understanding. The BD FACSymphony™ S6 Cell Sorter enables sorting of populations by using up to 50 parameters to define populations deep in your gating hierarchy. The detection technology in the BD FACSymphony™ A5 SE Cell Analyzer has enhanced resolution across the visible spectrum to identify and analyze rare cell types.
Learn more about the single-cell multiomics approach to comprehensively identify the gene and protein signature of T-cells.
The BD FACSymphony™ Cell Analyzers leverage the benefits of high-parameter flow cytometry for simultaneous measurements, depending on the system, of up to 50 different characteristics of a single cell, better accommodating your workflow needs. The BD FACSymphony™ A5 SE Cell Analyzer offers flexibility with an optical design that enables both spectral unmixing and compensation workflows. Our broad array of flow cytometry reagents including BD Horizon Brilliant™ Dyes, and BD Horizon RealYellow™ and BD Horizon RealBlue™ Fluorochromes with more than 9,000 human immunology research reagents can support cell therapy research. Made on demand BD OptiBuild™ Reagents also provide consistent performance and lot-to-lot consistency that can be tailored to your requirements. At your disposal is a wide range of custom products including BD® Abseq Reagents, instruments, conjugates, multicolor panels and cocktails to fit your needs.
BD products provide unique customizable end-to-end solutions with workflow flexibility for multiomic analysis and single-cell isolation of specific, rare cell populations. Led by you for your specific purpose, our team of scientists and experts can help with panel design, reagent selection, assay implementation, workflow optimization and experimental troubleshooting.
High-dimensional experiments result in more data to analyze, visualize and organize. Using the advanced computational tools within FlowJo™ Software, the leading software for flow cytometry analysis,* or data analysis pipelines, a tool providing visualization and tertiary analysis for single-cell multiomics analysis, you can unlock the insights from your data, with accessible features across a wide breadth of research applications. FlowJo™ Software enables flow cytometry data analysis with accessible features for immunophenotyping, cell cycle, proliferation, kinetics studies, quantitative population comparison, plate screening assays and more. Create publication worthy graphics using advanced algorithms through our plugin interface to analyze your high-dimensional data with the latest techniques to drive your research and discovery forward.
BD® Research Cloud, now available in the U.S., is a unique cloud-based ecosystem for the organization of and collaboration around flow cytometry experiments. The powerful software allows you to track experiments through customizable workflows, design new panels and create BD FACSDiva™ Software-based experiments away from the cytometer enabling efficiency, visibility and collaboration for your discovery work.
*In 2021, FlowJo™ Software was cited in leading immunology peer-reviewed journals 80% of the time a flow cytometry analysis software package was cited.
Flow cytometry sample preparation can be automated using the BD FACSLyric™ Flow Cytometer integrated with the BD FACSDuet™ Sample Preparation System. The sample preparation system is designed to complement and physically integrate with the flow cytometer to provide a complete end-to-end walkaway solution. This system helps streamline workflow and drive productivity while allowing for on-board automated antibody cocktail preparation, eliminating risk of errors due to manual pipetting. It supports different types of blood collection tubes and a wide variety of reagent vials delivering flexibility. The BD FACSLyric™ Flow Cytometer enables efficiency and productivity with automated laser alignment and compensation stability over 60 days and allows flexibility for acquisition using a universal loader with 21 different loading options including multiwell plates. The distinctive assay portability feature allows easy and efficient transferability of assays across sites.
Consistent reagent and instrument performance enables global collaboration
The BD FACSLyric™ Flow Cytometer has a unique assay portability feature that allows transfer of user-defined assays from one instrument to another, providing standardization and reproducible results across global manufacturing sites. This high-performance, highly sensitive flow cytometer demonstrates exceptional resolution and improved separation to make dim and rare populations easier to resolve. The BD FACSDuet™ Sample Preparation System provides accuracy and consistency in operation by minimizing manual intervention and an intuitive software interface minimizing training and variations due to different operators’ experience.
Custom ready-to-use multicolor reagents panels in liquid, dried, or lyophilized formats are customized to your specifications and requirements. Dried and lyophilized reagents are manufactured using proprietary BD Horizon™ Dri Chroma Reagent technology and BD Horizon™ Lyo Reagent technology, respectively and have extended stability compared to liquid reagents. All-in-one tube format reduces the need for manual pipetting and cocktailing which improves consistency, reduces operator errors and day-to-day variability and saves time. Certain preformulated dried reagent cocktails are available, like BD Horizon™ Dri TBNK + CD20 panel for cell therapy research workflows.
Functions in BD FACSuite™ Application assist in supporting 21 CFR Part 11 electronic record integrity features
Our flagship analyzer, BD FACSLyric™ Flow Cytometer is upgradeable to a 12-color configuration. It is powered by the BD FACSuite™ Application used for acquisition and analysis that helps to support 21 CFR Part 11 features with password protection, electronic signatures, automatic record keeping and audit trails. We provide operational qualification (OQ) by a BD field service engineer for features related to 21 CFR Part 11 features for the BD FACSLyric™ Flow Cytometer and the BD FACSuite™ Application. The BD FACSDuet™ System allows traceability of samples and worklists through carrier barcoding and supports ISO 15189-accredited labs with compliance with complete workflow traceability.
Global install base with guaranteed technical service support
With you in the driver’s seat and BD as your single partner for flow cytometry reagents, instruments and services you will benefits from synergies in user workflows and support provided. BD Technical Services provide installation qualification (IQ), operational qualification (OQ) and guaranteed technical service support to minimize instrument downtime. Explore our contract manufacturing offering of multicolor panels in liquid, dried or lyophilized formats to minimize errors and time associated with manual cocktailing of reagents. The BD FACSLyric™ Flow Cytometer has an install base of more than 3,000 units globally.
The BD Rhapsody™ HT Single-Cell Analysis System enables single-cell capture with reliable and reproducible results with minimal site-to-site variability making it suitable for translational, multi-center, multi-cohort studies. One readout can be used to analyze both protein and mRNA, reducing experimental time and increasing information derived from a single experiment. BD® AbSeq Antibody-Oligonucleotide Conjugates allows high-parameter protein expression data simultaneously with single-cell RNA-Seq data to significantly improve your understanding of individual cells. For example, targeted panels like the BD Rhapsody™ TCR/BCR Multiomic Assay Kit allow protein and whole transcriptome mRNA analysis to comprehensively understand immune cell function. Using genomic and proteomic information from single-cell analysis, you can focus on cells of interest for research and a deeper understanding. The BD FACSymphony™ S6 Cell Sorter enables sorting of populations by using up to 50 parameters to define populations deep in your gating hierarchy. The detection technology in the BD FACSymphony™ A5 SE Cell Analyzer has enhanced resolution across the visible spectrum to identify and analyze rare cell types.
Learn more about the single-cell multiomics approach to comprehensively identify the gene and protein signature of T-cells.
The BD FACSymphony™ Cell Analyzers leverage the benefits of high-parameter flow cytometry for simultaneous measurements, depending on the system, of up to 50 different characteristics of a single cell, better accommodating your workflow needs. The BD FACSymphony™ A5 SE Cell Analyzer offers flexibility with an optical design that enables both spectral unmixing and compensation workflows. Our broad array of flow cytometry reagents including BD Horizon Brilliant™ Dyes, and BD Horizon RealYellow™ and BD Horizon RealBlue™ Fluorochromes with more than 9,000 human immunology research reagents can support cell therapy research. Made on demand BD OptiBuild™ Reagents also provide consistent performance and lot-to-lot consistency that can be tailored to your requirements. At your disposal is a wide range of custom products including BD® Abseq Reagents, instruments, conjugates, multicolor panels and cocktails to fit your needs.
BD products provide unique customizable end-to-end solutions with workflow flexibility for multiomic analysis and single-cell isolation of specific, rare cell populations. Led by you for your specific purpose, our team of scientists and experts can help with panel design, reagent selection, assay implementation, workflow optimization and experimental troubleshooting.
High-dimensional experiments result in more data to analyze, visualize and organize. Using the advanced computational tools within FlowJo™ Software, the leading software for flow cytometry analysis,* or data analysis pipelines, a tool providing visualization and tertiary analysis for single-cell multiomics analysis, you can unlock the insights from your data, with accessible features across a wide breadth of research applications. FlowJo™ Software enables flow cytometry data analysis with accessible features for immunophenotyping, cell cycle, proliferation, kinetics studies, quantitative population comparison, plate screening assays and more. Create publication worthy graphics using advanced algorithms through our plugin interface to analyze your high-dimensional data with the latest techniques to drive your research and discovery forward.
BD® Research Cloud, now available in the U.S., is a unique cloud-based ecosystem for the organization of and collaboration around flow cytometry experiments. The powerful software allows you to track experiments through customizable workflows, design new panels and create BD FACSDiva™ Software-based experiments away from the cytometer enabling efficiency, visibility and collaboration for your discovery work.
*In 2021, FlowJo™ Software was cited in leading immunology peer-reviewed journals 80% of the time a flow cytometry analysis software package was cited.
Flow cytometry sample preparation can be automated using the BD FACSLyric™ Flow Cytometer integrated with the BD FACSDuet™ Sample Preparation System. The sample preparation system is designed to complement and physically integrate with the flow cytometer to provide a complete end-to-end walkaway solution. This system helps streamline workflow and drive productivity while allowing for on-board automated antibody cocktail preparation, eliminating risk of errors due to manual pipetting. It supports different types of blood collection tubes and a wide variety of reagent vials delivering flexibility. The BD FACSLyric™ Flow Cytometer enables efficiency and productivity with automated laser alignment and compensation stability over 60 days and allows flexibility for acquisition using a universal loader with 21 different loading options including multiwell plates. The distinctive assay portability feature allows easy and efficient transferability of assays across sites.
Consistent reagent and instrument performance enables global collaboration
The BD FACSLyric™ Flow Cytometer has a unique assay portability feature that allows transfer of user-defined assays from one instrument to another, providing standardization and reproducible results across global manufacturing sites. This high-performance, highly sensitive flow cytometer demonstrates exceptional resolution and improved separation to make dim and rare populations easier to resolve. The BD FACSDuet™ Sample Preparation System provides accuracy and consistency in operation by minimizing manual intervention and an intuitive software interface minimizing training and variations due to different operators’ experience.
Custom ready-to-use multicolor reagents panels in liquid, dried, or lyophilized formats are customized to your specifications and requirements. Dried and lyophilized reagents are manufactured using proprietary BD Horizon™ Dri Chroma Reagent technology and BD Horizon™ Lyo Reagent technology, respectively and have extended stability compared to liquid reagents. All-in-one tube format reduces the need for manual pipetting and cocktailing which improves consistency, reduces operator errors and day-to-day variability and saves time. Certain preformulated dried reagent cocktails are available, like BD Horizon™ Dri TBNK + CD20 panel for cell therapy research workflows.
Functions in BD FACSuite™ Application assist in supporting 21 CFR Part 11 electronic record integrity features
Our flagship analyzer, BD FACSLyric™ Flow Cytometer is upgradeable to a 12-color configuration. It is powered by the BD FACSuite™ Application used for acquisition and analysis that helps to support 21 CFR Part 11 features with password protection, electronic signatures, automatic record keeping and audit trails. We provide operational qualification (OQ) by a BD field service engineer for features related to 21 CFR Part 11 features for the BD FACSLyric™ Flow Cytometer and the BD FACSuite™ Application. The BD FACSDuet™ System allows traceability of samples and worklists through carrier barcoding and supports ISO 15189-accredited labs with compliance with complete workflow traceability.
Global install base with guaranteed technical service support
With you in the driver’s seat and BD as your single partner for flow cytometry reagents, instruments and services you will benefits from synergies in user workflows and support provided. BD Technical Services provide installation qualification (IQ), operational qualification (OQ) and guaranteed technical service support to minimize instrument downtime. Explore our contract manufacturing offering of multicolor panels in liquid, dried or lyophilized formats to minimize errors and time associated with manual cocktailing of reagents. The BD FACSLyric™ Flow Cytometer has an install base of more than 3,000 units globally.