-
Reagents
- Flow Cytometry Reagents
-
Western Blotting and Molecular Reagents
- Immunoassay Reagents
-
Single-Cell Multiomics Reagents
- Accessories
- BD® AbSeq Assay
- BD Rhapsody™ Accessory Kits
- BD® Single-Cell Multiplexing Kit
- BD Rhapsody™ TCR/BCR Next Multiomic Assays
- BD Rhapsody™ Targeted mRNA Kits
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit
- BD Rhapsody™ TCR/BCR Profiling Assays
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
-
Functional Assays
-
Microscopy and Imaging Reagents
-
Cell Preparation and Separation Reagents
- Dehydrated Culture Media
Old Browser
Request a Quote/Inquiry
Please fill in the following information and we will get in touch with you regarding your query.
개요
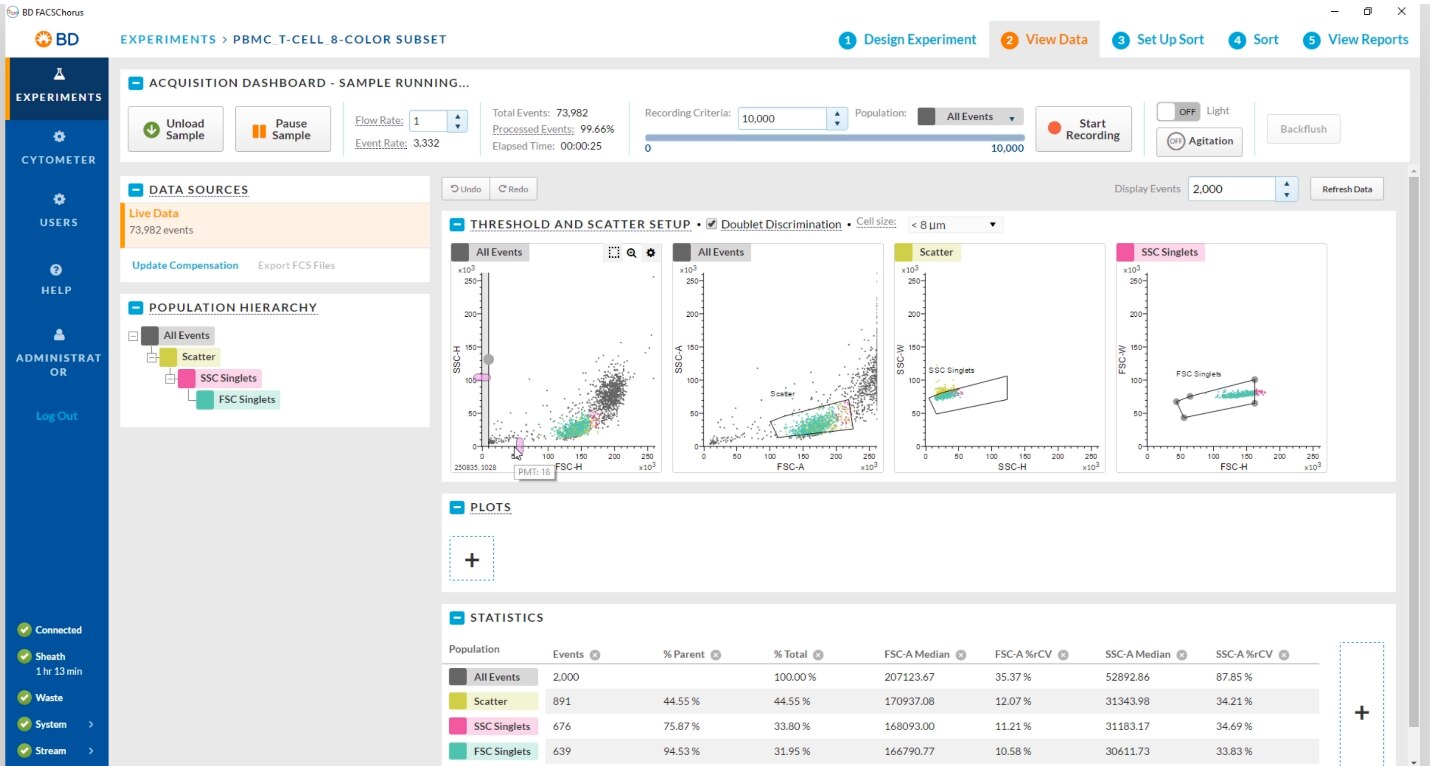
BD FACSChorus ™ Software는 과학적 연구에 집중할 수 있도록 수동 설정 및 모니터링이 필요 없는 간소화된 워크플로우를 사용하여 분류(sorting)를 단순화합니다.
이 소프트웨어는 처리량을 개선하고 가동중지시간을 줄이는 고급 자동화 기술을 이용하여 전체 세포분류 프로세스를 안내하고 실험을 성공적으로 이끕니다.
특징
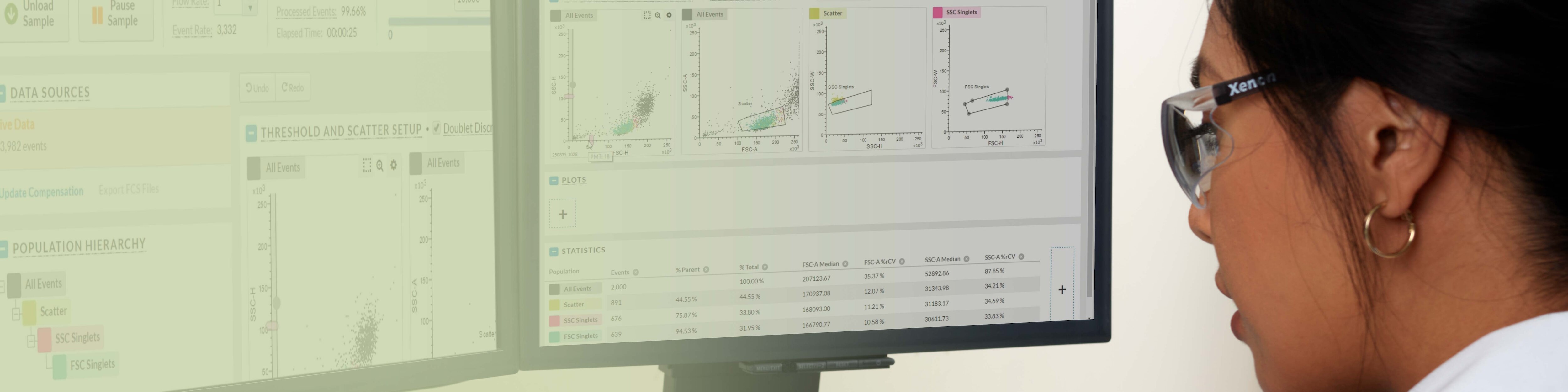
BD FACSChorus ™ 소프트웨어 설계는 직관적이고 사용하기 쉽기 때문에 초보 사용자도 분류(sorting) 작업에 빠르게 적응할 수 있습니다.

기능:
- Laser delay, drop delay, side stream의 자동설정
- 워크플로우를 안내하는 인터페이스
- 화면에 표시되는 지침
- 인덱스 정렬 획득 및 분석
- 디폴트 plot 및 sort gate
- 튜브overflow방지
- 작업자 안전 보호를 위한 deflection plate(필요한 경우에만) 자동켜짐
- FCS 3.1 호환성
- 입증된 BD FACS™ Accudrop 및 BD® Sweet Spot 기술
응용분야
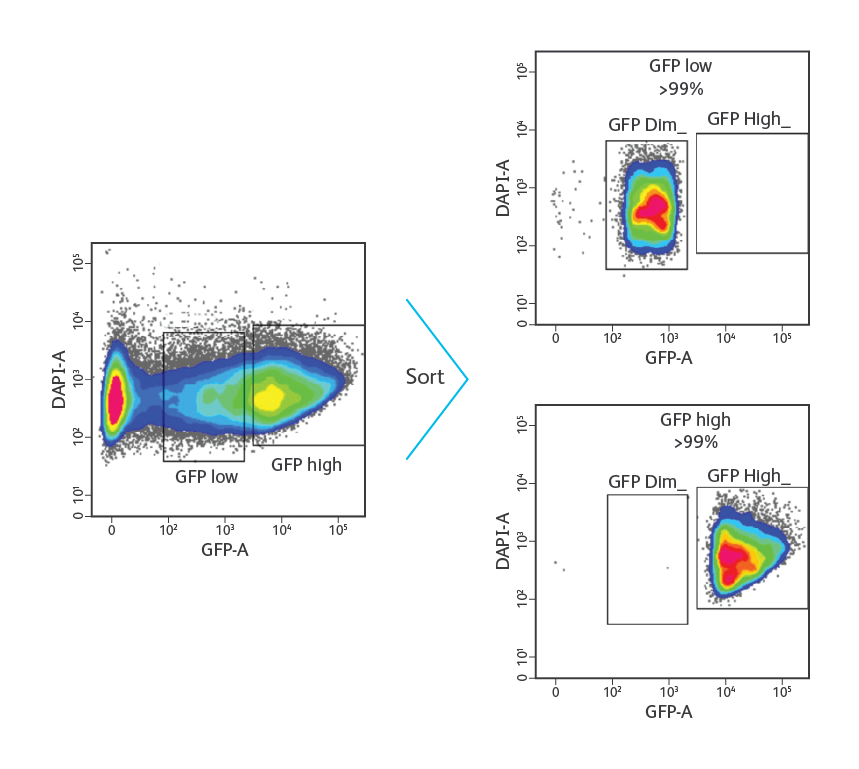
HEK-293 세포를 0.25 μg의 pAcGFP (Clontech) 벡터로 transfection시키고 GFP의 발현에 대해 분석했습니다. 다양한 수준의 GFP를 발현하는 세포는 명확하게 구분 될 수 있습니다. DAPI는 죽은 세포를 배제하기 위해 사용되었습니다. Purity 모드에서 초당 1,000 개 event에서 낮은 수준과 높은 수준의 GFP를 발현하는 세포를 식별하고 분류하기 위해 gate를 그렸습니다. 정렬 후 분석결과 두 집단 모두에서 순도가 99 %를 초과하는 것으로 나타났습니다.
HEK-293 세포를 70 % 에탄올로 고정하고 0.5μg / mL의 DAPI로 염색했습니다.
(A) 집단 계층. SSC, FSC 및 DAPI에 대한 A, H 및 W parameter를 순차적으로 사용하여 doublet 및 세포 응집체를 제외했습니다. 이 실험에 사용된 gating 전략은 유사 분열 수치를 과소 평가하거나 배제했을 가능성이 있습니다.
(B) DNA content 분석. DAPI singlet gate 내의 세포는 2N과 4N 사이의 DNA content를 보여주어> 4N doublet 또는 응집체의 배제를 확인했습니다.
(C) Single-cell 증착 정렬 후 검증. DAPI singlet gate의 세포는 99.7 %의 single-cells증착 효율로 3 개의 96 -multiwell plates로 분류되었습니다. 현미경을 사용하여 well당 하나의 세포가 존재함을 확인했습니다. 하나의 세포를 포함하는well의 대표 이미지를 표시했습니다.

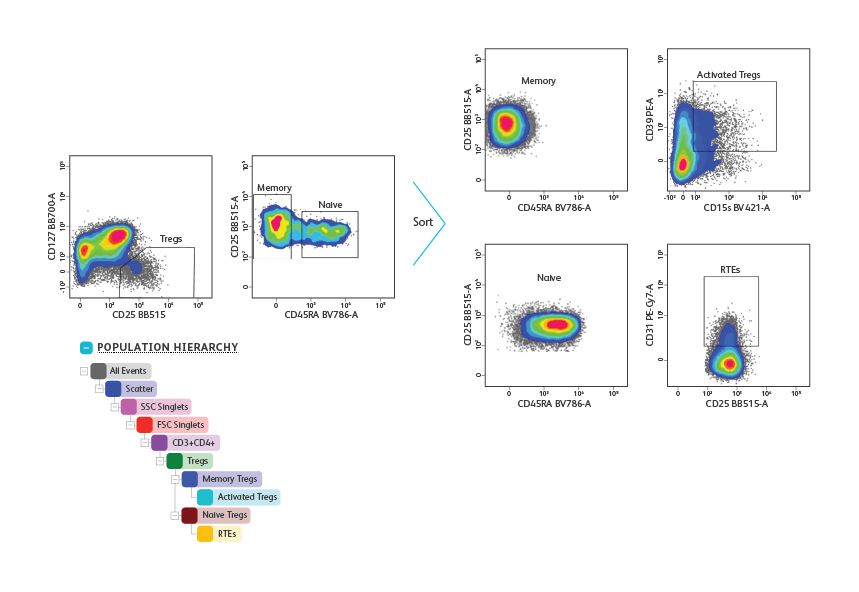
건강한 기증자로부터 얻은 말초 혈액 단핵 세포를 분리하고 Treg subset 의 측정 및 정제를 위해 표면 marker cocktail(CD3, CD4, CD25, CD127 및 CD45RA)로 염색했습니다. 림프구 및 singlet은 우선 FSC,SSC 특성을 기반으로 gating 된 다음 CD3 + CD4 + T cell로 gating(표시되지 않음) 했습니다. Tregs 는CD127low/neg CD25high로 식별되었습니다. Treg gate에서 CD45RA + naïve 및 CD45RA– memory Tregs를 식별하고 purity 모드에서 초당 5,000 개 이벤트로 분류했습니다. 정렬 후 분석 결과 memory와 naïve Treg의 동종 집단이 밝혀졌습니다. 이어서 정제된 세포를 면역 표현형을 위한 추가 표면 marker(CD31, CD39 및 CD15)에 대해 염색했습니다. CD31 + recent thymic emigrants (RTEs)은 CD45RA + naive Tregs 내에서 측정된 반면, 고도로 활성화된 Tregs는 CD45RA- memory Tregs 내에서 측정되었습니다.
BALB/c 마우스 비장 세포는 collagenase를 사용한 기계적 mincing 및 효소적 소화 후에 분리되었습니다.
(A) BD IMag™ Mouse Dendritic Cell Enrichment Set 를 bulk 선택에 사용하여 MHC II + CD11chigh 기존 수지상 세포의 CD8α + 및 CD8α- subset를 각각 35 배 및 29 배 농축했습니다.
(B) Bulk 선택 후, CD8α + 및 CD8α- subset는 purity 모드에서 초당 1,000 개 event로 분류되었습니다. 정렬 후 분석 결과 두 subset에서 모두 순도가 99 %를 초과하는 것으로 나타났습니다.

-
BD FACSChorus™ FC Bead Lot Updater
연구용입니다. 진단용 또는 치료용으로 사용할 수 없습니다.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.